28
Oct 25
Received ANDA Approval for Ticagrelor Tablets, 60 mg and 90 mg.
Read More
16
Oct 25
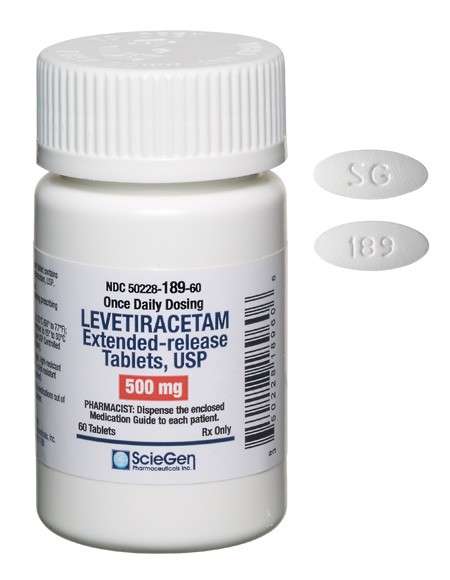
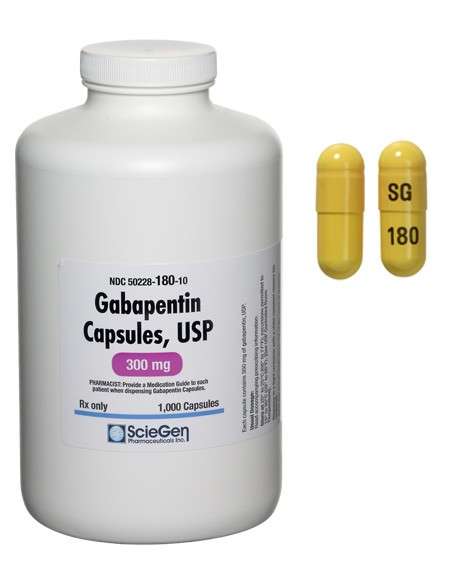
Received ANDA Approval for Gabapentin Tablets (Once-Daily), 300 mg and 600 mg.
Read More
6
Aug 25
Received ANDA Approval for Rivaroxaban Tablets, USP 2.5 mg, 5 mg, 10 mg and 20 mg
Read More
12
Mar 25
Received ANDA Tentative Approval for Tentative ANDA approval for Apixaban Tablets, 2.5mg and 5mg.
Read More
6
Mar 25
Received ANDA Tentative Approval for Sitagliptin Tablets, USP 25mg, 50mg, 100mg
Read More
16
Oct 23
Received ANDA Approval for Clotrimazole Topical Solution USP, 1%
Read More
11
Oct 23
Received ANDA Approval for Naproxen and Esomeprazole Magnesium Delayed-Release Tablets 375mg/20 mg and 500mg/20mg.
Read More
28
Jul 23
Received ANDA Approval for Phytonadione Tablets USP, 5 mg.
Read More
6
Apr 23
Received ANDA Approval for Diltiazem Hydrochloride Extended-Release Tablets, 120 mg, 180 mg, 240 mg, 300 mg, 360 mg,
and 420 mg
Read More
3
Oct 22
Received ANDA Approval for Diltiazem Hydrochloride Tablets USP, 30 mg, 60 mg, 90 mg, and 120 mg.
Read More

News / Announcements
28
Oct 25
Received ANDA Approval for Ticagrelor Tablets, 60 mg and 90 mg.
Read More
16
Oct 25
Received ANDA Approval for Gabapentin Tablets (Once-Daily), 300 mg and 600 mg.
Read More
6
Aug 25
Received ANDA Approval for Rivaroxaban Tablets, USP 2.5 mg, 5 mg, 10 mg and 20 mg
Read More
12
Mar 25
Received ANDA Tentative Approval for Tentative ANDA approval for Apixaban Tablets, 2.5mg and 5mg.
Read More
6
Mar 25
Received ANDA Tentative Approval for Sitagliptin Tablets, USP 25mg, 50mg, 100mg
Read More
16
Oct 23
Received ANDA Approval for Clotrimazole Topical Solution USP, 1%
Read More
11
Oct 23
Received ANDA Approval for Naproxen and Esomeprazole Magnesium Delayed-Release Tablets 375mg/20 mg and 500mg/20mg.
Read More
28
Jul 23
Received ANDA Approval for Phytonadione Tablets USP, 5 mg.
Read More
6
Apr 23
Received ANDA Approval for Diltiazem Hydrochloride Extended-Release Tablets, 120 mg, 180 mg, 240 mg, 300 mg, 360 mg,
and 420 mg
Read More
3
Oct 22
Received ANDA Approval for Diltiazem Hydrochloride Tablets USP, 30 mg, 60 mg, 90 mg, and 120 mg.
Read More