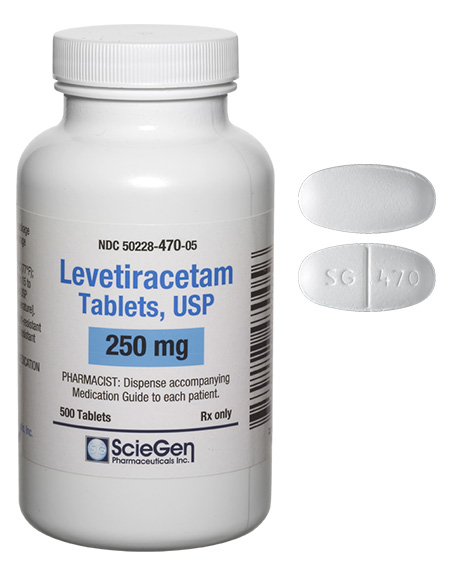
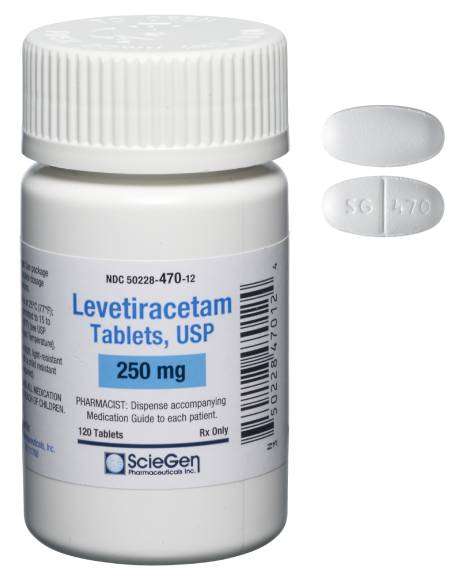
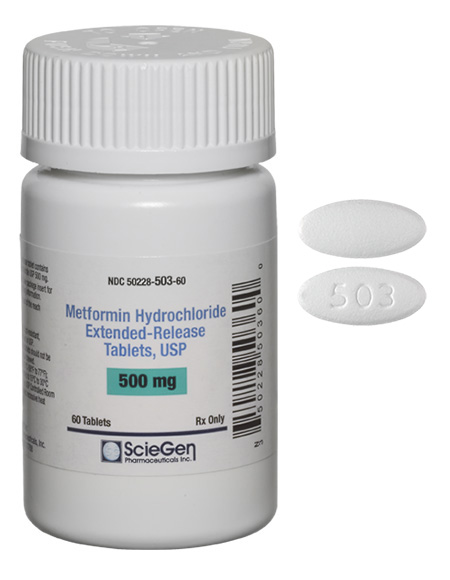
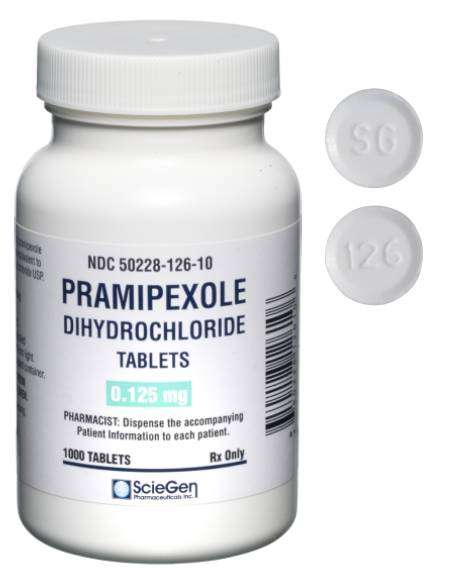
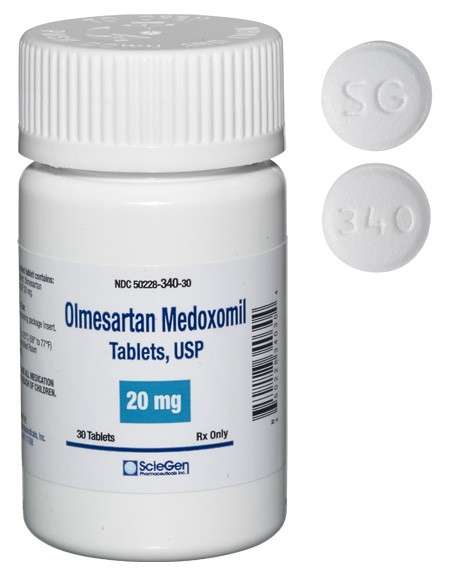
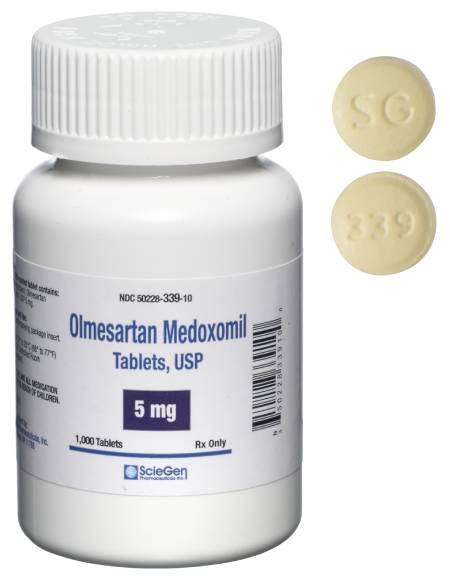
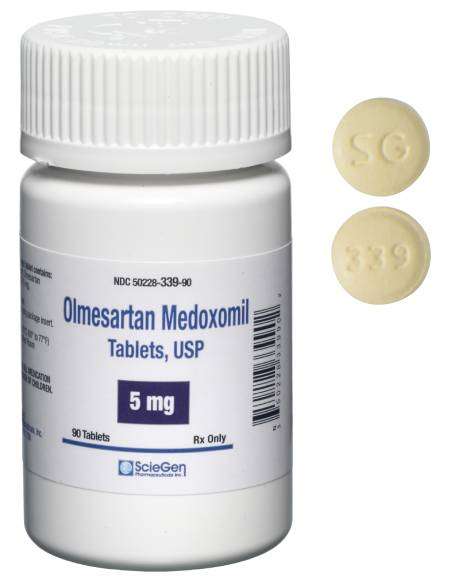
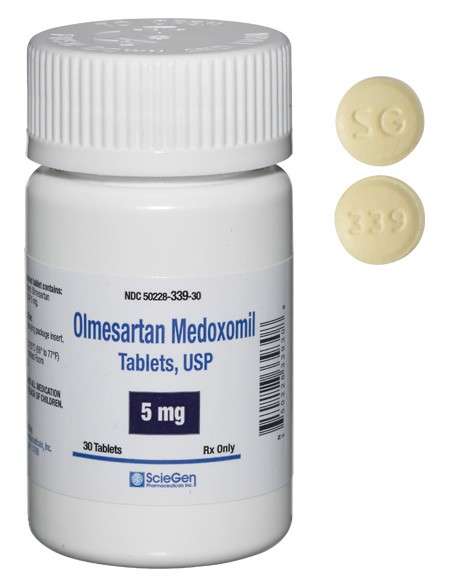
generic pharmaceutical products.




Welcome to
ScieGen Pharmaceuticals
ScieGen Pharmaceuticals, Inc is a fast-growing generic pharmaceutical company located in Hauppauge, NY. Our core business is in the areas of Development, Manufacturing, Marketing, and Distribution of finished dose generic pharmaceutical products. ScieGen has a robust product development pipeline with over 60 ANDA’s filed, and more in process. We aim to provide patients with high quality and cost effective generic medicines.
All ScieGen products are manufactured in the United States of America, at our state of the art cGMP facilities in Hauppauge, New York.

We aim to provide healthcare at economical prices to make this a healthier world to live in.
News / Announcements
From 2017 ScieGen is producing this product and distributed into the market.
During this period we never received any complaints from the customers on any adverse events and side effects. ScieGen performed Bioequivalence studies at Synchron (a Contract Research organization, who performs the Bioavailability and Bioequivalence studies).
Very recently FDA found some data integrity issues at Synchron. The issues observed at Synchron are not related to ScieGen’s product.
As precautionary measure FDA asked all ANDA holders who performed their respective BE studies at Synchron for last 10 years to repeat the Biostudies and submit the data with a agency with 12 months. As part of this FDA decided to change status of all ANDA holders to “BX”, accordingly Sciegen’s product rating changed. FDA not asked us to recall the product. FDA has stated in its press release and webpage that “FDA has been conducting an investigation of postmarket safety reports for marketing applications that relied on bioequivalence studies conducted by Synchron or Panexcell for approval. To date, the agency has not identified signals related to problems with the safety or quality of the approved drugs. FDA remains vigilant and if the agency finds safety issues, we will take action and provide the latest information as appropriate. Patients should not make changes to their treatment except in consultation with their health care professional.”
ScieGen is planned to perform the BE studies, and will submit the data to agency by May’2022.
Please visit the FDA webpage https://www.fda.gov/drugs/drug-safety-and-availability/notification-pharmaceutical-companies-clinical-and-bioanalytical-studies-conducted-panexcell
ScieGen Metformin ER tablets 500 mg and 1000 mg complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
NDMA testing with a validated test method is a part of release and stability testing in Metformin ER tablets manufactured by ScieGen.
Sciegen Pharmaceuticals Inc states that the Immediate release tablets Metformin Hydrochloride Tablets, USP 500 mg, 850 mg & 1000 mg manufactured by Sciegen Pharmaceuticals Inc are not part of recall regarding Metformin products.
Sciegen Metformin IR tablets complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
Several batches manufactured by Sciegen are tested with a validated test method and the levels are found to ‘below detectable limit’.
This data has been submitted to FDA.
It should also be noted that FDA has stated in its press release on May 28th ‘The FDA recommends that health care professionals continue to prescribe metformin when clinically appropriate; FDA testing has not shown NDMA in immediate release (IR) metformin products (the most commonly prescribed type of metformin).’
https://www.fda.gov/news-events/press-announcements/fda-alerts-patients-and-health-care-professionals-nitrosamine-impurity-findings-certain-metformin
Management
Dr. Pailla Malla Reddy (CEO) is a successful pharmaceutical
entrepreneur, he got abundant experience in various positions and managed pharmaceutical companies in Lagos, Nigeria, India and USA. Dr Reddy founded Sciegen Pharmaceuticals Inc. in 2009. Sciegen operates a facility in Hauppuage, NY. Dr Reddy has successfully launched and actively manages other leading pharmaceutical companies in the USA and other countries as a Director.
He also founded Bactolac Pharmaceutical, Inc. in 1995 and has grown the company exponentially over the years. He also operates other companies…