16
Apr 21
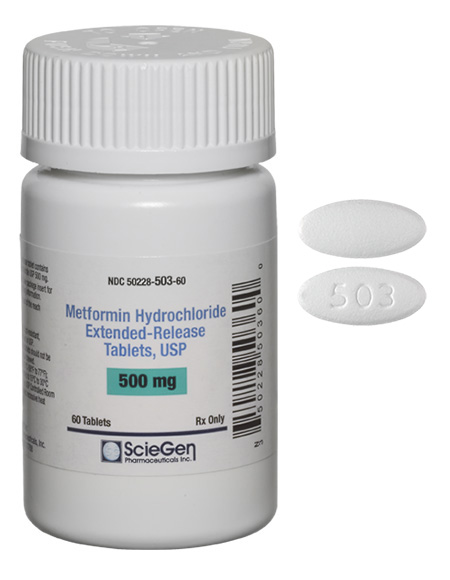
ScieGen receives approval for Metformin ER tablets 500 mg and 1000 mg [ RLD , Glumetza ]
ScieGen Metformin ER tablets 500 mg and 1000 mg complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
NDMA testing with a validated test method is a part of release and stability testing in Metformin ER tablets manufactured by ScieGen.
Read More
ScieGen Metformin ER tablets 500 mg and 1000 mg complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
NDMA testing with a validated test method is a part of release and stability testing in Metformin ER tablets manufactured by ScieGen.
23
Feb 21
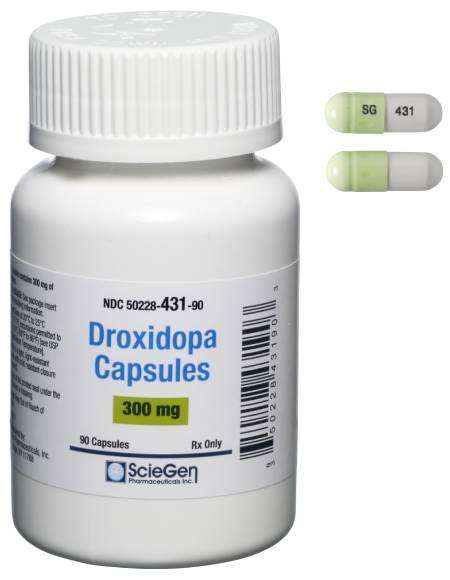
Received ANDA Approval for Droxidopa Capsules, 100 mg, 200 mg, and 300 mg
Read More
26
Jan 21
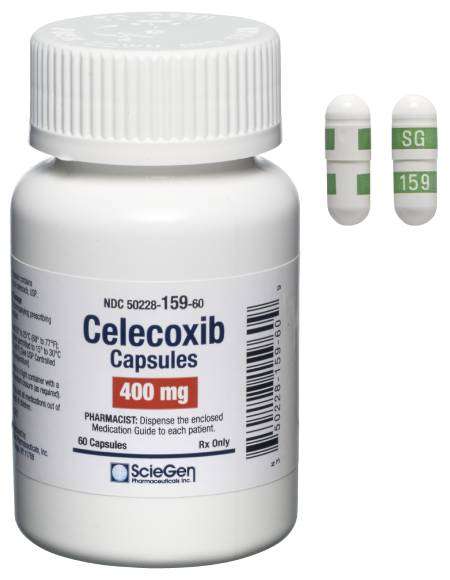
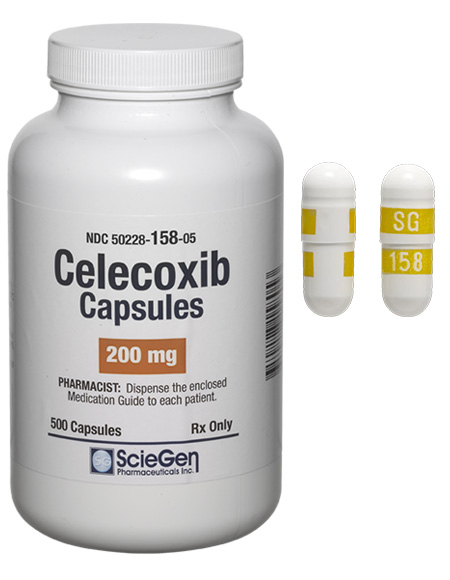
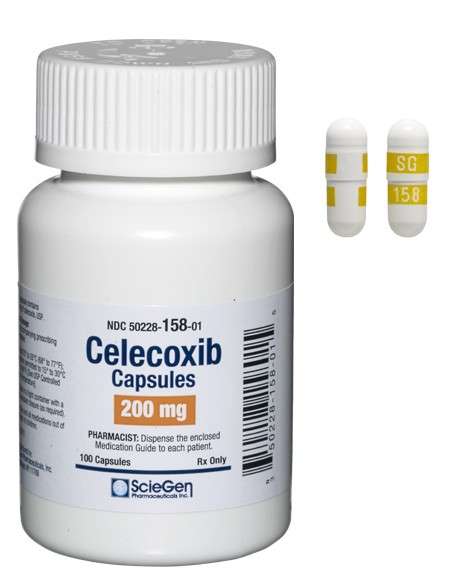
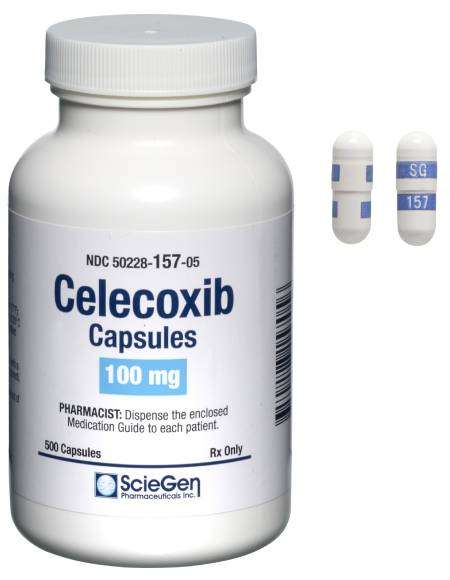
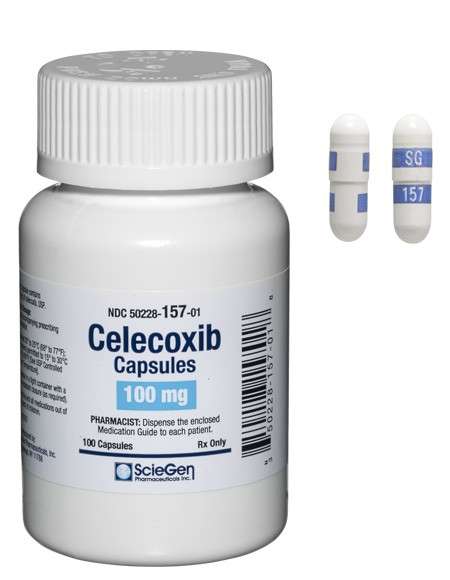
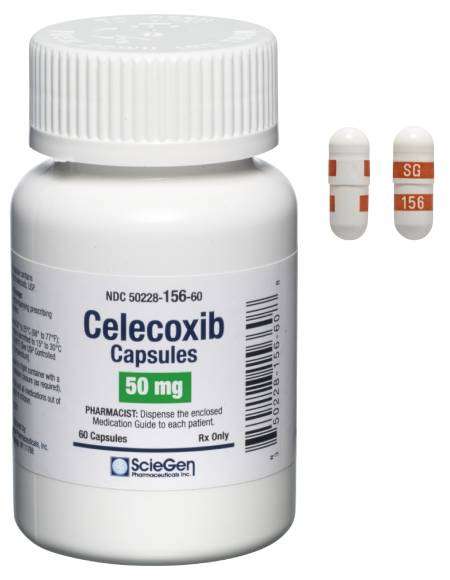
Received ANDA Approval for Celecoxib Capsules, 50 mg, 100 mg, 200 mg, and 400 mg
Read More
5
Jan 21
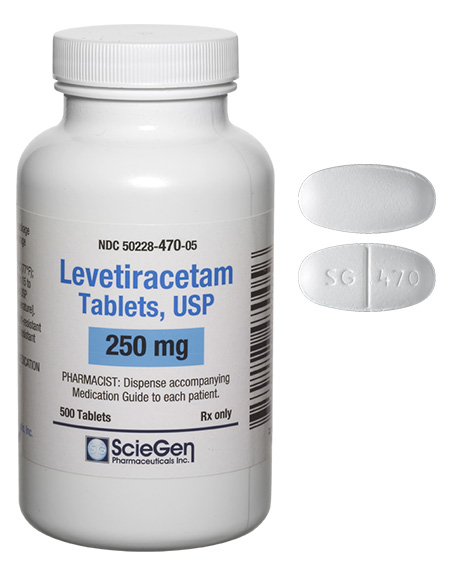
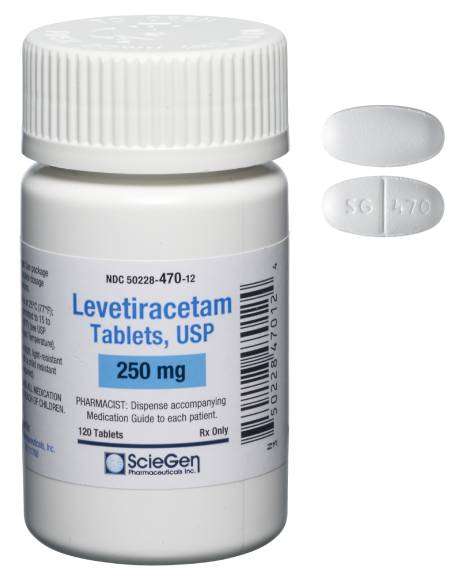
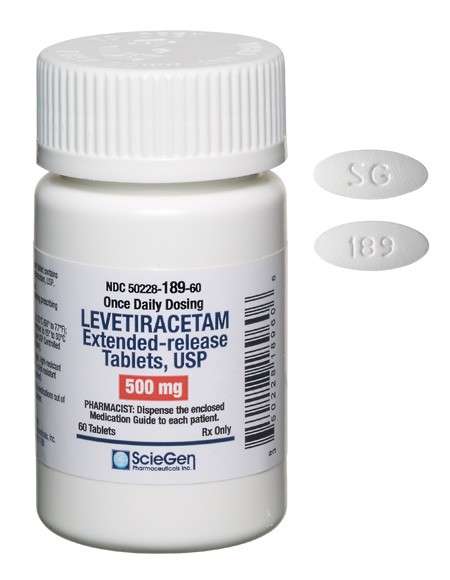
Received ANDA Approval for Levetiracetam Extended-Release Tablets USP, 500 mg and 750 mg.
Read More
23
Dec 20
Received ANDA Tentative Approval for Pirfenidone Tablets, 267 mg and 801 mg
Read More
14
Dec 20
Received ANDA Tentative Approval for Pirfenidone Capsules, 267 mg
Read More
7
Dec 20
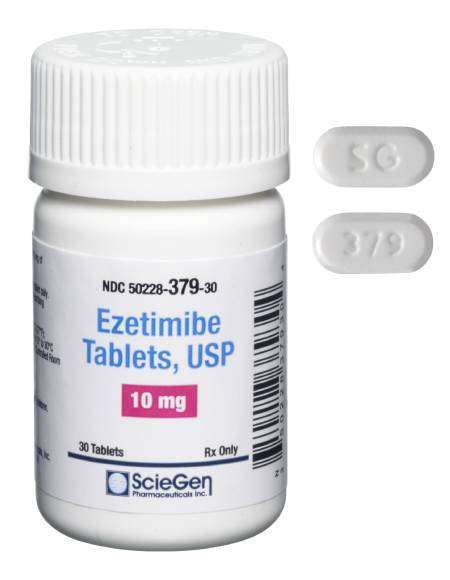
Received ANDA Approval for Ezetimibe Tablets, 10mg
Read More
18
Nov 20
Received ANDA Tentative Approval for Lacosamide Tablets, 50 mg, 100 mg, 150 mg, and 200 mg
Read More
14
Aug 20
Sciegen Metformin product – not part of Recall
Sciegen Pharmaceuticals Inc states that the Immediate release tablets Metformin Hydrochloride Tablets, USP 500 mg, 850 mg & 1000 mg manufactured by Sciegen Pharmaceuticals Inc are not part of recall regarding Metformin products.
Sciegen Metformin IR tablets complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
Several batches manufactured by Sciegen are tested with a validated test method and the levels are found to ‘below detectable limit’.
This data has been submitted to FDA.
Read More
Sciegen Pharmaceuticals Inc states that the Immediate release tablets Metformin Hydrochloride Tablets, USP 500 mg, 850 mg & 1000 mg manufactured by Sciegen Pharmaceuticals Inc are not part of recall regarding Metformin products.
Sciegen Metformin IR tablets complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
Several batches manufactured by Sciegen are tested with a validated test method and the levels are found to ‘below detectable limit’.
This data has been submitted to FDA.
28
May 20
It should also be noted that FDA has stated in its press release on May 28th ‘The FDA recommends that health care professionals continue to prescribe metformin when clinically appropriate;
FDA testing has not shown NDMA in immediate release (IR) metformin products (the most commonly prescribed type of metformin).’
https://www.fda.gov/news-events/press-announcements/fda-alerts-patients-and-health-care-professionals-nitrosamine-impurity-findings-certain-metformin
Read More
https://www.fda.gov/news-events/press-announcements/fda-alerts-patients-and-health-care-professionals-nitrosamine-impurity-findings-certain-metformin

News / Announcements
16
Apr 21
ScieGen receives approval for Metformin ER tablets 500 mg and 1000 mg [ RLD , Glumetza ]
ScieGen Metformin ER tablets 500 mg and 1000 mg complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
NDMA testing with a validated test method is a part of release and stability testing in Metformin ER tablets manufactured by ScieGen.
Read More
ScieGen Metformin ER tablets 500 mg and 1000 mg complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
NDMA testing with a validated test method is a part of release and stability testing in Metformin ER tablets manufactured by ScieGen.
23
Feb 21
Received ANDA Approval for Droxidopa Capsules, 100 mg, 200 mg, and 300 mg
Read More
26
Jan 21
Received ANDA Approval for Celecoxib Capsules, 50 mg, 100 mg, 200 mg, and 400 mg
Read More
5
Jan 21
Received ANDA Approval for Levetiracetam Extended-Release Tablets USP, 500 mg and 750 mg.
Read More
23
Dec 20
Received ANDA Tentative Approval for Pirfenidone Tablets, 267 mg and 801 mg
Read More
14
Dec 20
Received ANDA Tentative Approval for Pirfenidone Capsules, 267 mg
Read More
7
Dec 20
Received ANDA Approval for Ezetimibe Tablets, 10mg
Read More
18
Nov 20
Received ANDA Tentative Approval for Lacosamide Tablets, 50 mg, 100 mg, 150 mg, and 200 mg
Read More
14
Aug 20
Sciegen Metformin product – not part of Recall
Sciegen Pharmaceuticals Inc states that the Immediate release tablets Metformin Hydrochloride Tablets, USP 500 mg, 850 mg & 1000 mg manufactured by Sciegen Pharmaceuticals Inc are not part of recall regarding Metformin products.
Sciegen Metformin IR tablets complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
Several batches manufactured by Sciegen are tested with a validated test method and the levels are found to ‘below detectable limit’.
This data has been submitted to FDA.
Read More
Sciegen Pharmaceuticals Inc states that the Immediate release tablets Metformin Hydrochloride Tablets, USP 500 mg, 850 mg & 1000 mg manufactured by Sciegen Pharmaceuticals Inc are not part of recall regarding Metformin products.
Sciegen Metformin IR tablets complying to FDA requirements of NDMA content less 96 nanograms (0.0376 ppm).
Several batches manufactured by Sciegen are tested with a validated test method and the levels are found to ‘below detectable limit’.
This data has been submitted to FDA.
28
May 20
It should also be noted that FDA has stated in its press release on May 28th ‘The FDA recommends that health care professionals continue to prescribe metformin when clinically appropriate;
FDA testing has not shown NDMA in immediate release (IR) metformin products (the most commonly prescribed type of metformin).’
https://www.fda.gov/news-events/press-announcements/fda-alerts-patients-and-health-care-professionals-nitrosamine-impurity-findings-certain-metformin
Read More
https://www.fda.gov/news-events/press-announcements/fda-alerts-patients-and-health-care-professionals-nitrosamine-impurity-findings-certain-metformin